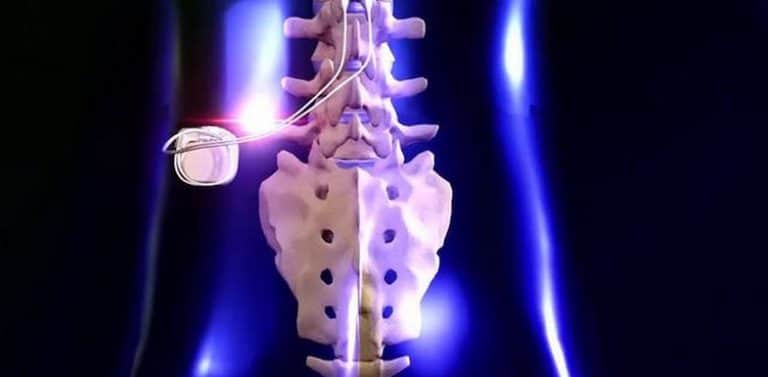
The procedure is usually safe, but may be associated with certain risks including change in electrode position, breakage, battery failure, bleeding, infection and development of tolerance.ĭorsal column stimulators do not treat the underlying cause of pain, but with its use, the intensity, frequency and duration of pain are usually reduced, lowering the need for pain medication and sometimes surgery. Full Body MRI Systems WaveWriter Alpha SCS Systems. Spinal cord stimulator placement is a non-destructive, reversible procedure and thus is often an attractive alternative for patients who have failed other treatment and surgical options.

Here you will find important information about ImageReady MRI technology, supported devices and conditions for use. Spinal cord stimulation (SCS) is a proven long-term and effective therapy for managing chronic pain. the dorsal columns of the spinal cord in order to block pain sensations. What are the Associated Risks and Complications of Dorsal Column Stimulator? ImageReady MRI Spinal Cord Stimulator Systems allow patients to safely undergo MRI scans. Electrical impulses are generated at the pulse generator by remote control and are transmitted to the electrodes. The following two mechanisms have been considered.
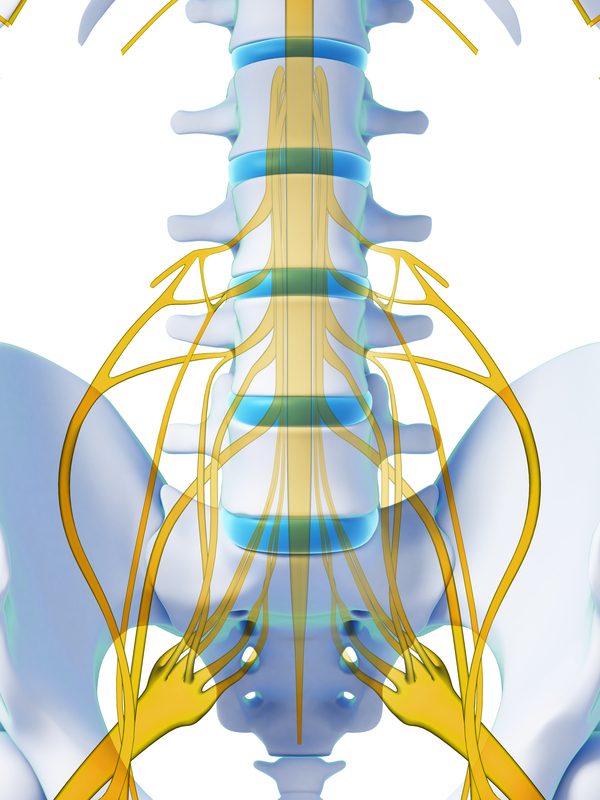
However, the mechanism underlying its analgesic effect has not been fully elucidated. The remaining components are connected externally. Dorsal column stimulation is a minimally invasive treatment for intractable pain in the back and extremities. SCS is a treatment method that alleviates pain by applying a weak electrical stimulation to the dorsal column of the spinal cord. for the treatment of CRPS I and causalgia (CRPS II) of the. This is done either surgically or through the skin under radiographic guidance. The only FDA-approved dorsal root ganglion (DRG) technology. Is this guidance up to date We reviewed the evidence in February 2014.We found nothing new that affects the recommendations in this guidance. Depending on the location of your pain, the electrodes are placed in the neck, upper or lower back region of the spinal cord. Evidence-based recommendations on spinal cord stimulation for chronic pain of neuropathic or ischaemic origin in adults. whether the patient is likely to derive a significant therapeutic benefit from continuing use of an implanted nerve stimulator within a trial period of four weeks. How is the Procedure of Dorsal Column Stimulator Performed?Ī dorsal column stimulator consists of electrodes, a battery-operated pulse generator, a remote control device and connecting wires. Refer to the Local Coverage Article: Billing and Coding: Spinal Cord Stimulation (Dorsal Column Stimulation), A57023, for all coding information. DCS acts either by blocking the pain impulse pathway or by stimulating the production of endorphins (chemical substances produced by the body that decrease the sensation of pain). Dorsal column stimulation (DCS), also known as spinal cord stimulation (SCS), is a neuromodulation therapy that works by applying an electrical current to the spinal cord at the source of the pain. It is used to treat chronic disabling pain such as failed back surgery syndrome and complex regional pain syndrome, which show little or no improvement with other treatments such as medication or surgery. The dorsal column stimulator (DCS) or spinal column stimulator is a device that relieves chronic pain associated with spinal nerves through electrical stimulation.
